
Loftware Cloud is the world’s first public, validation-ready Cloud labeling solution. It is the simplest way to design and print labels in a regulated environment.

Click the flashing orange dot below to move through the steps to see how Loftware can work for you.

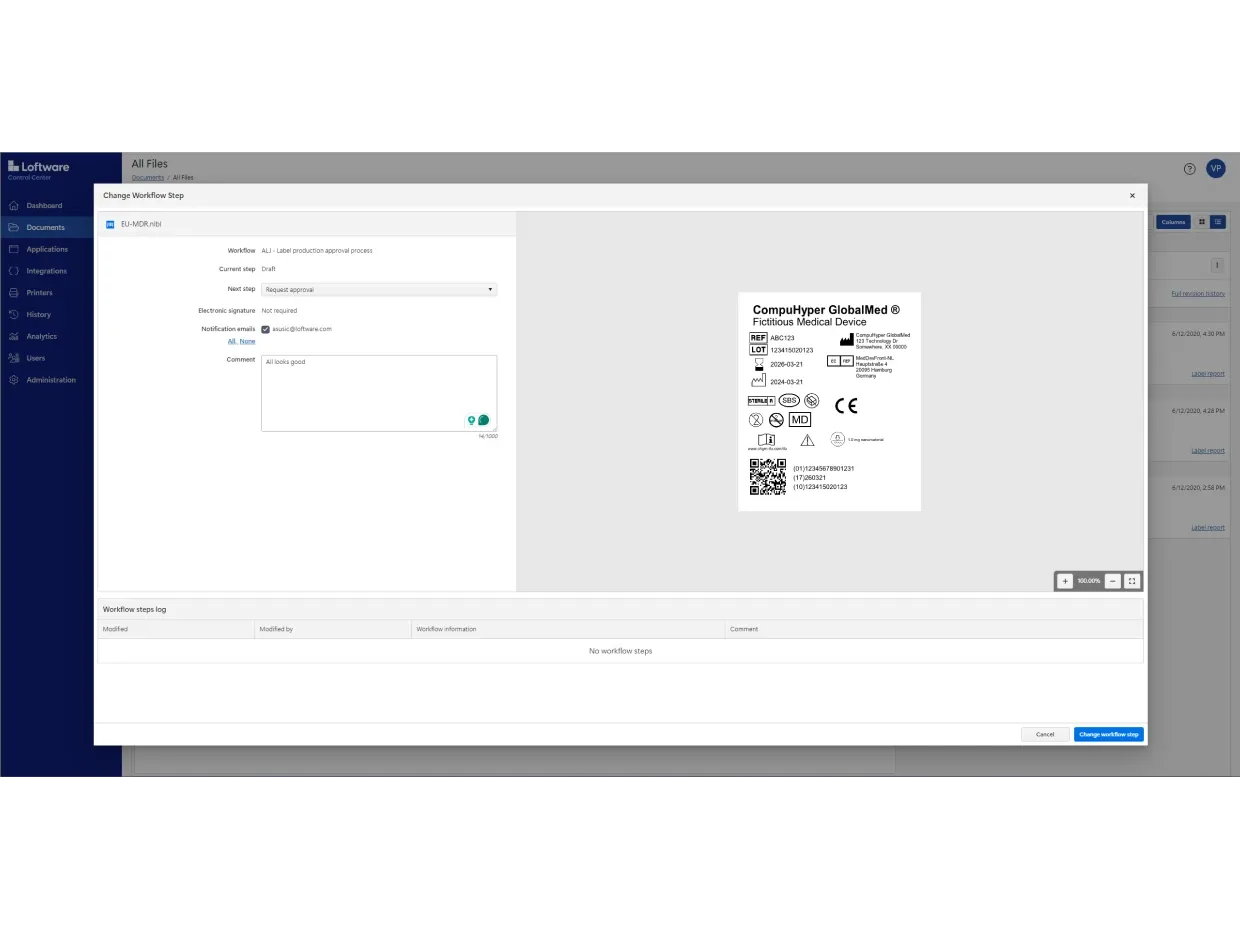
Use Loftware Cloud to digitize your entire approval process. You can also automate mass label changes and approvals without creating hundreds of label variations.
您可以使用系统的三层环境对变更继续测试,然后再将它们应用到生产环境中。这三层环境是:开发 (DEV)、质量保证 (QA) 和生产 (PROD)。
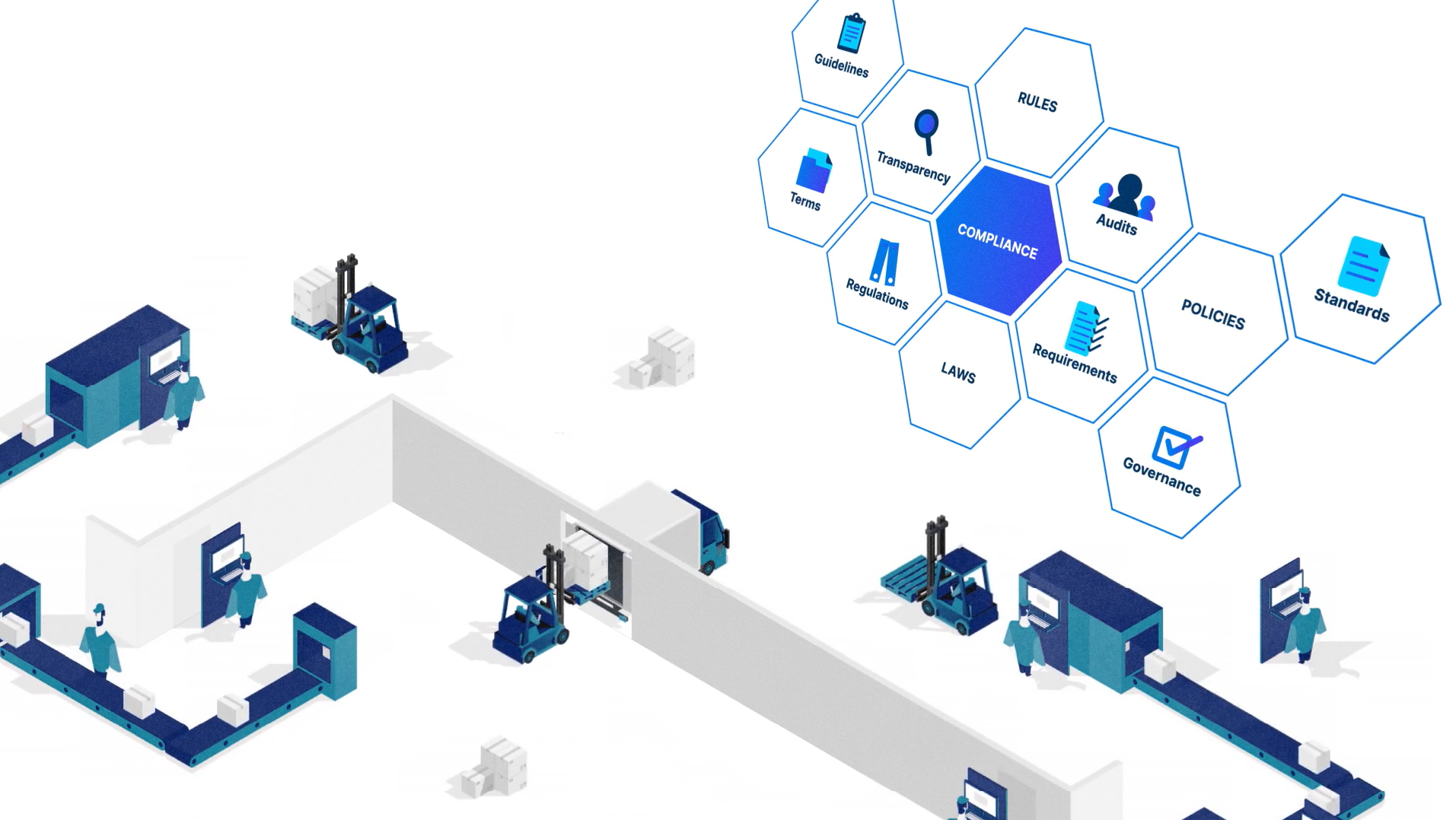
Loftware Cloud is designed to comply with the major regulations impacting the life science industry, including EU MDR, FDA UDI, FDA 21 CFR Part 11 and EU GMP Annex 11.
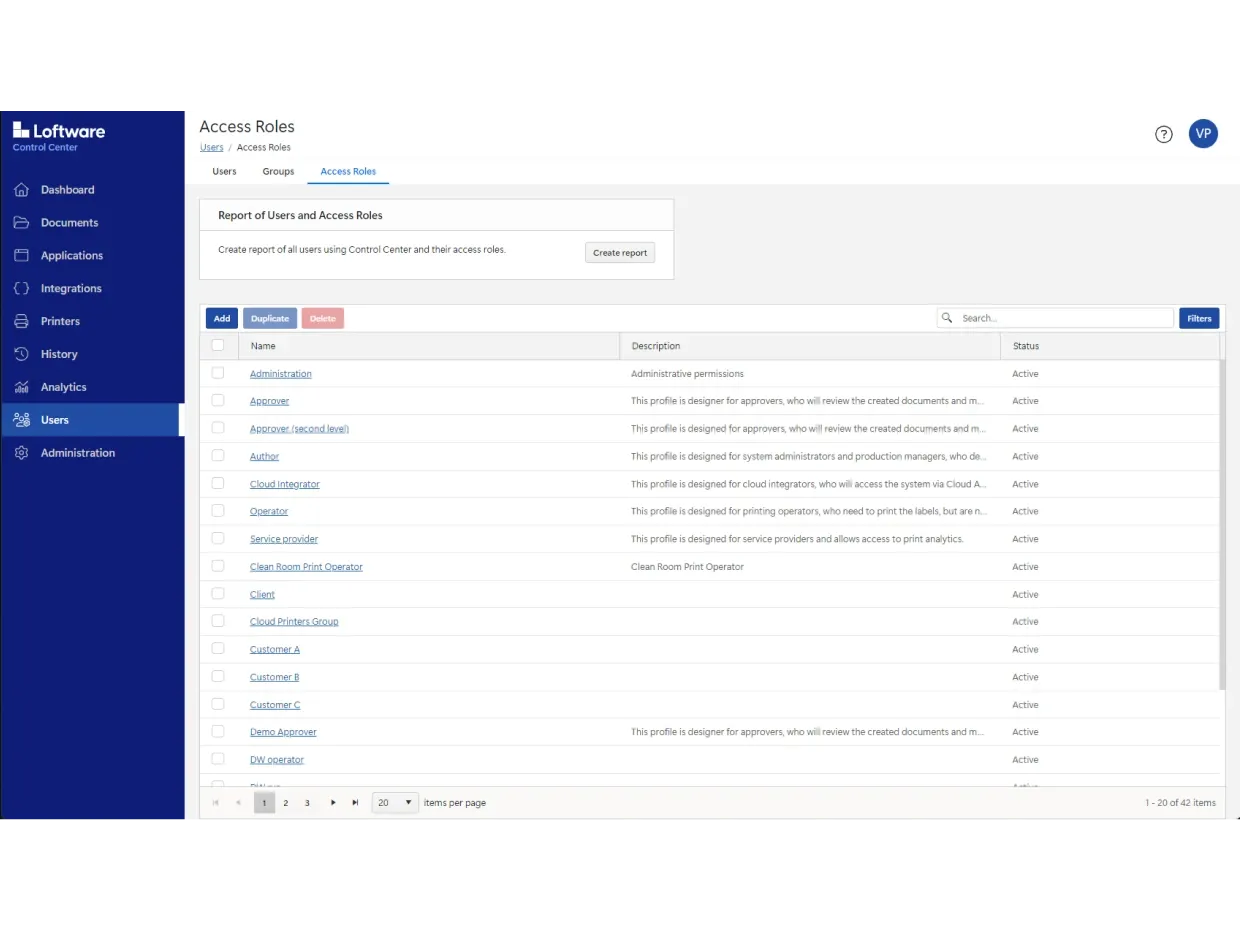
它包含使用系统内置的基于角色的访问权限控制、文档版本控制、可定制的审批流程、电子记录和电子签名(ERES)。
您还将获得 12 年的打印历史记录,该记录使您可以直观地跟踪在此期间打印的每个标签。

Loftware Cloud is a validation-ready labeling solution. It is designed to streamline the validation process, which is further enhanced with the option of the Validation Acceleration Pack (VAP).
Our team can assist with IQ, OQ and PQ documentation as well. We also reduce the validation burden by only updating the software once a year. And when it’s time for the software release, we give you a three-month window before updating your production environment.
